CenBRAIN members design and implement state-of-the-art optical biosensors, electronic biosensors and microfluidic lab-on-chips to tackle various challenges in the area of wearable and implantable medical devices and precision medicine (Figure 1).

Figure 1. Wearable and implantable electronics for health management and continuous drug monitoring.
Biosensors detect biomolecules and have important applications in many areas including biomedicine, food safety, biosafety, and environment. We are introducing label-free optical biosensors based on a CMOS compatible nanomaterial-porous silicon. It is a nanostructured material with large surface area and tunable morphology, making it very suitable for constructing biosensors. In addition, we exploit Localized Surface Plasmon Resonance (LSPR) with resonant optical porous silicon devices to achieve high sensitivity of biomolecular detection (Figure 2 a, b, c). Gold nano thin film is deposited on the biosensor surface to form resonant mode and is modified by bio-receptors to capture biomolecules. The binding of target virus and receptors on the biochip surface can change the refractive index of sample solution, and then a shift of biosensor reflectance spectrum can be observed. This change is instantaneous, and therefore, the response time of proposed biosensors can be very fast. Meanwhile, the biochip is highly compact in structure, which helps miniaturizing the size of biosensing system and can be applied to a variety of commercial devices.
In addition, we achieved an automatic detection system that can realize automatic sample loading, detection, and data processing as shown in Figure 2d.

Figure 2. Fabrication of porous silicon: (a) Six-inch wafer; (b) PDMS well array in 96-well format, and an enlarged view of a single well; (c) SARS-CoV-2 binding on biosensor surface; (d) Automatic detection system.
We are now moving into an era of advanced healthcare in which wearable and implantable biochips will automatically adjust drug dosing in response to patient health conditions, greatly enabling the management of diseases and enhancing individualized therapy [1]. Continuous therapeutic drug monitoring (TDM) has the great potential to revolutionize the precision medicine by providing real-time information about an individual's response to therapeutics, while on-site TDM, which is more often implemented at the current stage, enables physicians to make rapid treatment decisions to achieve maximal drug efficacy. To achieve on-site TDM, we experiment fiber optic-based biolayer interferometry (FO-BLI) biosensors to detect therapeutic biologicals and small molecule drugs for the treatment of neurodegenerative diseases in minutes [2]. To achieve continuous TDM, we are devoted to building microfluidic-based electrochemical biosensors for recording drugs in seconds in blood. The sensing elements can be monoclonal antibodies, molecular imprint polymers [3], or specific aptamers.
Human brain achieves amazing complex functions that machine cannot do, such as emotions and recognition. In this respect, AI and neural network technologies will never replace human brain despite their rapid development. To have deeper insights into the brain’s mechanism, we are devoted to building biological neural network with the use of neural cells [4]. In this network, individual cells are captured on a microelectrode array via a manipulation layer and once captured, the activities of single cell will be recorded in real time following stimulation via a microelectrode that holds it (Figure 3a). A smart gas supply system is integrated to maintain a stable microenvironment for long-term culture of cells without losing their activities. Besides, we are building microfluidic-based miniaturized microchips for testing of neurotransmitters. These electrochemical microchips in matrices format are extracellular fluids (Figure 3b).
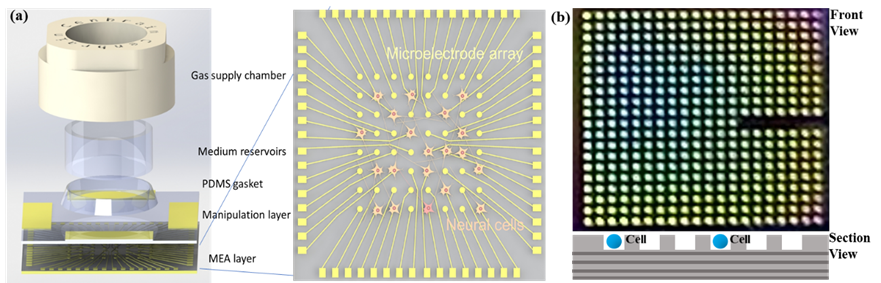
Figure 3. (a) Biological neural network, and (b) Microfluidic biochip.
Precise monitoring of SARS-CoV-2 antibodies, and in particular neutralizing ones (NAbs), is of particular importance when evaluating the efficacy of new vaccines and therapeutic biologicals to treat COVID-19 during clinical trials. To achieve on-site sample-to-answer, we propose a fiber optic-based biolayer interferometry (FO-BLI) biosensor with the use of an effective signal enhancer for rapid, sensitive and automated detection of NAbs in human sera within 10 mins [5]. The FO-BLI biosensor uses an optic fiber as the substrate to allow competitive binding interaction to happen between the NAbs, the SARS-CoV-2 protein and the coronavirus receptor (Figure 4). In particular, (a) when no NAbs exist, the binding between receptor binding domain-horseradish peroxidase (RBD-HRP) and angiotensin-converting enzyme 2 receptor (ACE2) contributes to highest detection signals; while (b) the presence of NAbs blocks HRP-conjugated RBD from binding to immobilized ACE2 protein. In this bioassay, a metal precipitate was used to amplify the optical signals. Direct monitoring of SARS-CoV-2 binding antibodies is even more convenient using the introduced FO-BLI system. Moreover, we evaluate dried blood spots to replace invasive venous blood as an easy and alternative sample matric for high-through analysis of NAbs in larger-scale sero-surveillance [6].
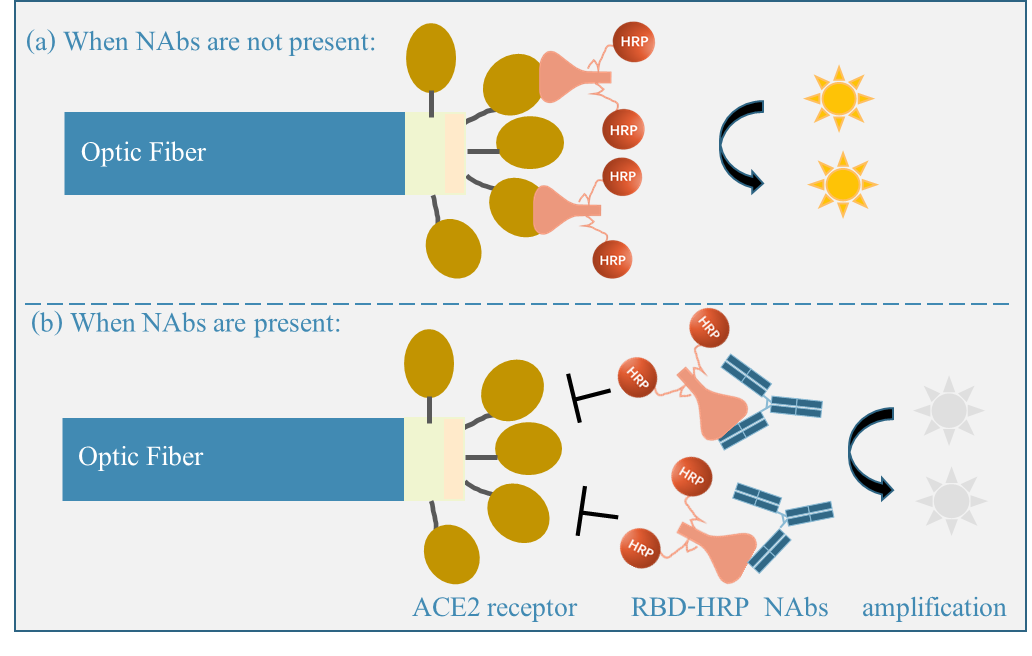
Figure 4. Optical fiber-based SARS-CoV-2 NAbs detection principle.
Wearable biosensors have received tremendous attention over the past decade owing to their great potential in predictive analytics and treatment toward personalized medicine. Flexible electronics could serve as an ideal platform for personalized wearable devices because of their unique properties such as light weight, low cost, high flexibility and great conformability. Unlike most reported flexible sensors that mainly track physical activities and vital signs, the new generation of wearable and flexible chemical sensors enables real-time, continuous and fast detection of accessible biomarkers from the human body and allows for the recording of large-scale information about the individual’s dynamic health status at the molecular level. We are implementing analytical methods that can achieve real-time monitoring in sweat and develop more efficient sweat sampling methods to resolve sweat evaporation, contamination from skin.
[1] S. Bian, B. Zhu, G. Rong, and M. Sawan, "Towards wearable and implantable continuous drug monitoring: A review," Journal of Pharmaceutical Analysis, vol. 11, no. 1, pp. 1-14, 2021, doi: 10.1016/j.jpha.2020.08.001.
[2] S. Bian, Y. Tao, and M. Sawan, "Rapid, highly-sensitive biosensing carbamazepine in whole blood of epileptic patients," Submitted.
[3] A. Hammoud, D. Chhin, D. K. Nguyen, and M. Sawan, "A new molecular imprinted PEDOT glassy carbon electrode for carbamazepine detection," Biosensors and Bioelectronics, vol. 180, p. 113089, 2021, doi: 10.1016/j.bios.2021.113089.
[4] H. Zhang, G. Rong, S. Bian, and M. Sawan, “Lab-on-Chip Microsystems for Ex Vivo Network of Neurons Studies: A Review,” Frontiers in Bioengineering and Biotechnology, vol. 10, 2022-February-16, 2022.
[5] S. Bian, M. Shang, and M. Sawan, “Rapid biosensing SARS-CoV-2 antibodies in vaccinated healthy donors,” Biosensors and Bioelectronics, vol. 204, pp. 114054, 2022/05/15/, 2022.
[6] S. Bian et al., "Evaluating an easy sampling method using dried blood spots to determine vedolizumab concentrations," Journal of Pharmaceutical and Biomedical Analysis, vol. 185, p. 113224, 2020, doi: 10.1016/j.jpba.2020.113224.