Type 2 diabetes (T2D), a chronic metabolic disorder affecting hundreds of millions worldwide, significantly diminishes patients' quality of life. Conventional management strategies, including medication injections and continuous glucose monitoring, often fall short of delivering dynamic, real-time control.
In response to this challenge, closed-loop neuromodulation has emerged as a promising frontier. By continuously monitoring physiological signals and delivering precise stimulation on demand, this technology offers a pathway toward intelligent, personalized diabetes care.
Recently, the CenBRAIN Neurotech at Westlake University achieved a breakthrough in diabetes treatment technology. Our research center proposed and validated a prototype implantable closed-loop neuromodulation system for managing type 2 diabetes. This system innovatively utilizes pancreatic electrophysiological signals to simultaneously achieve sensing and therapeutic functions, laying a solid experimental and technical foundation for truly personalized and automated glycemic control.
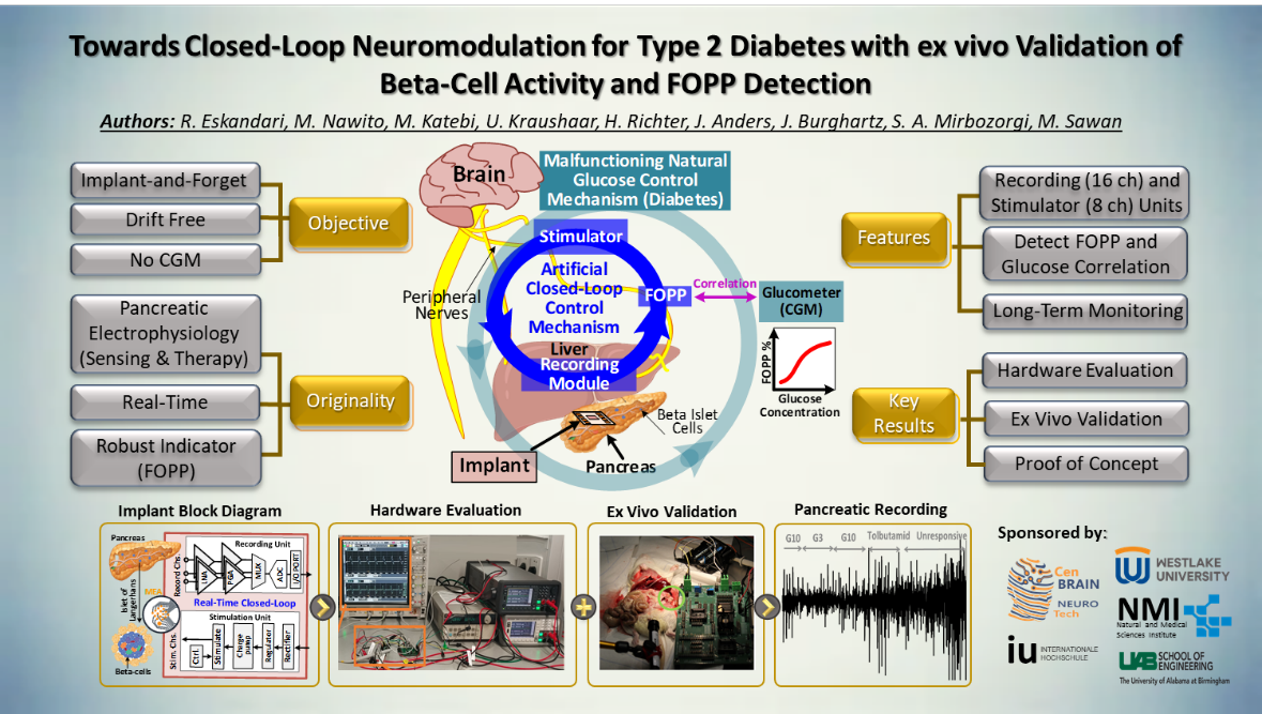
Fig. 1. Graphical Abstract.
This international collaborative contribution is published in the IEEE Transactions on Biomedical Circuits and Systems (IEEE TBioCAS), a leading journal in the field. Dr. Razieh Eskandari, Research Assistant Professor at CenBRAIN Neurotech Center of Excellence, is the first author, with Chair Professor Mohamad Sawan serving as corresponding author. Westlake University is the primary institution.
ABSTRACT
We present in this contribution an implantable closed-loop neuromodulation prototype system for type 2 diabetes (T2D) management, which leverages pancreatic electrophysiology as both a sensing and therapeutic modality.
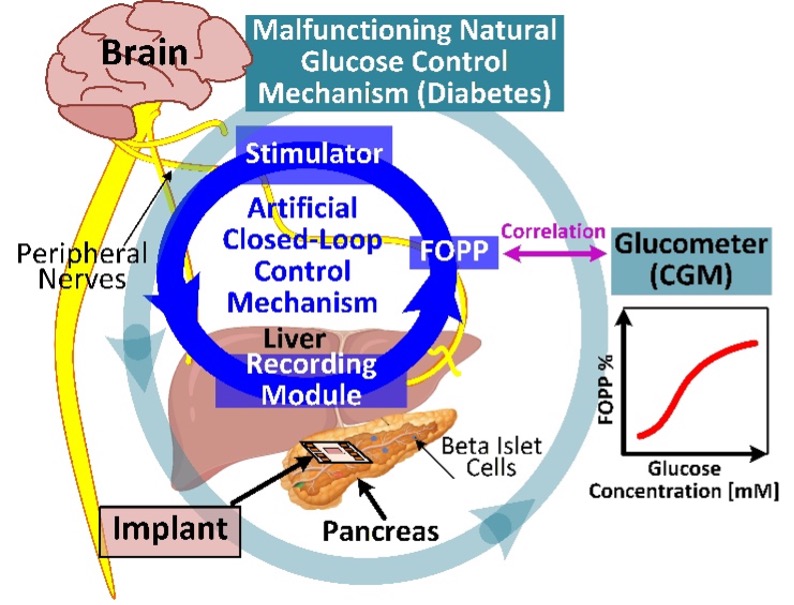
Fig. 2. Concept of the proposed diabetes management closed-loop neuromodulator.
Among candidate biomarkers, the fraction of plateau phase (FOPP) emerges as a robust indicator of glucose dynamics. Hence, the neural interface is optimized for low-power measurement of the electrical activity of the beta-cells with high accuracy in direct readout mode and long-term monitoring in FOPP mode.
The experimental framework was established using a perfused pancreas model, first in mice and then optimized for rats, with glucose-dependent signals captured via a custom 16-channel neural interface. Results confirmed the feasibility of extracting FOPP in ex vivo settings, though signal complexity differed from isolated islets in vitro.

Fig. 3. Key features, evaluation methods, and the pancreatic recording.
Additionally, a designed, implemented and fabricated 8-channel electrical stimulator with adjustable current levels and optimized charge balancing technique, demonstrated the capability to meet physiological requirements for beta-cell activation. While integration of AI-based classifiers for advanced FOPP–glucose correlation remains a future step, this study establishes the foundational experimental and technological evidence for a next-generation closed-loop neuromodulator.
More Information
ESKANDARI R., NAWITO M., KATEBI M., KRAUSHAAR U., RICHER H., ANDERS J., BUGHARTZ J., MIRBOZIRGI A., SAWAN M., “Towards Closed-Loop Neuromodulation for Type 2 Diabetes with ex vivo Validation of Beta-Cell Activity and FOPP Detection”, IEEE Transactions on Biomedical Circuits and Systems, Online, 2025.
https://pubmed.ncbi.nlm.nih.gov/41379895/
This manuscript is an extended version of our previously awarded ISCAS conference paper, with substantial new technical content and experimental validation. The work represents a strong international collaboration between Westlake University (China), the University of Applied Sciences Bad Reichenhall (Germany), NMI Naturwissenschaftliches und Medizinisches Institut an der Universität Tübingen (Germany), and the University of Alabama at Birmingham (USA), bringing together complementary expertise in circuit design, biomedical systems, and experimental validation.